(주)에스켐텍
콜로이달실리카Colloidal Silica
01 입자 및 표면상태
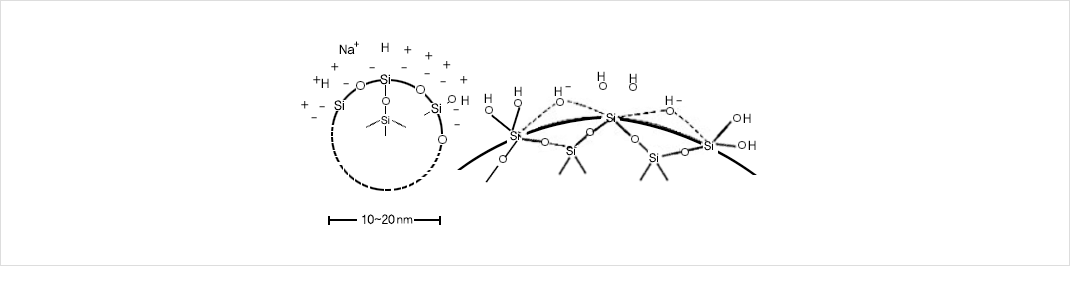
- 입자의 구조는 구형이며 수중에서 콜로이드 상태로 분산되어있다.
- 표면에 Si-OH와 OH-이온들이 존재하여 음전하를 형성한다.
- 알칼리 이온인 Na+이온에 의해 전기적 이중층이 형성되어, 입자간의 반발에 의해 안정화 되어있음.
- 이 전하밸런스가 무너지는 경우 분자끼리 서로 결합하여 응고, 겔화, 응집현상이 발생.
- 표면에 Si-OH와 OH-이온들이 존재하여 음전하를 형성한다.
- 알칼리 이온인 Na+이온에 의해 전기적 이중층이 형성되어, 입자간의 반발에 의해 안정화 되어있음.
- 이 전하밸런스가 무너지는 경우 분자끼리 서로 결합하여 응고, 겔화, 응집현상이 발생.
02 pH의 영향
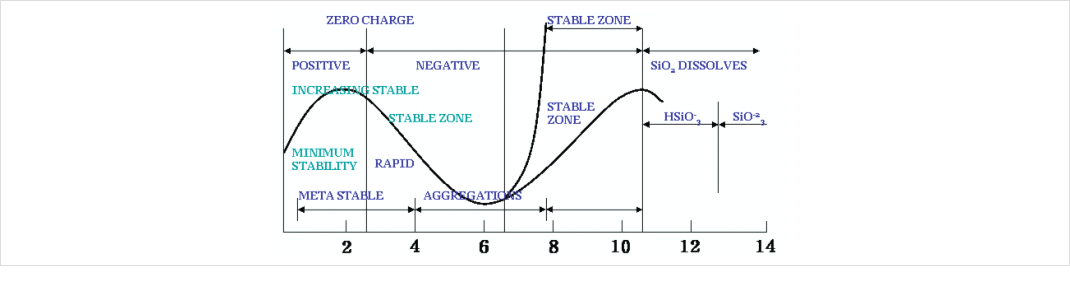
콜로이달 실리카의 경우 pH에 따라 안정성이 달라진다. 그래프에서 보면 pH 2의 경우 등전점으로
준 안정한 상태를 나타내며 자사제품 중 SS-SOL 30A가 이에 해당한다. 또한 알칼리인 경우 pH 8~10
사이인 경우 안정한 상태를 나타내는데 자사의 SS-SOL시리즈중 알칼리성을 나타내는 SS-SOL 30,
SS-SOL 30F 등 대부분의 염기성 졸이 이에 해당된다.
*유효기간 : 제품 라벨지에 표기
*유효기간 : 제품 라벨지에 표기
03 SS-SOL의 용도
SS-SOL은 안정성이 우수하여 장기간 보존할 수 있고, 아래와 같은 특성을 지니므로 이를 잘 활용하여 다방면에서 공업적 용도로 사용할 수 있습니다.
-
마찰계수의 증대
고체표면에 부착된 실리카 입자에 의해 마찰계수를 증대시킨다. -
부착, 피막
고체 표면으로의 강하고 단단한 부착 및 충진 피막의 형성을 행한다. -
경화성
소결에 의해 단단한 겔을 형성하므로 분말소재의 경화,성형 시 사용이 가능하다. -
겔화 반응에 의한 점성증대
겔화반응에 의해 점성을 증대시키고, 균일한 겔을 형성하며 분말체를 균일하게 분산시킬 수 있다. 또한 지지체를 안정화 시킬 수 있다 -
친수성 향상, 대전방지
Si-OH기로 인한 친수성 향상 및 대전을 방지할 수 있다. -
반응성의 증대
이온의 농도가 낮고 순도가 높은 실리카 원료이므로, 액상혼합시 반응이 균일하게 진행되며, 표면적이 크므로 반응속도를 향상시킨다. -
큰 표면적
콜로이드 상태의 실리카 분자로 큰 표면적을 및 균일한 세공을 가지는 실리카겔을 얻을 수 있다. -
함침제 및 충전제
다공성 물질의 충진에 사용이 가능하다. Na농도가 적으므로 환경친화적으로 사용가능하다.
04 섬유

-
양털과 모직물 제조
유연제에 SS-SOL 30을 희석하여 첨가하므로써 유연작용 및 인장력을 늘려서 섬유의 파손을 방지할 수 있습니다. -
수지가공
유연제에 SS-SOL 30을 희석하여 첨가하므로써 유연작용 및 인장력을 늘려서 섬유의 파손을 방지할 수 있습니다. -
사이징
SS-SOL을 사이징에 이용하면 아교의 분산이 고루고루 이루어지며 건조시간이 단축되고 응집력이 증대됩니다. -
나일론 어망제조
SS-SOL을 사용하면 색깔의 변형을 막고 색안정성을 향상하며, 비틀림을 방지한다. 취급간수를 용이하게 하며, 수명연장과 내구성 강화를 향상시킵니다. -
옷감
SS-SOL과 알루미나 졸을 혼합하여 염색 후 모사를 담궈 건조시킴으로 좋은 효과를 얻을 수 있습니다.
05 제지공정

-
DIAZO 감광지
DIAZO 감광지에 SS-SOL을 첨가하여 용지의 표면을 평편하고 미끄럽게 하여 사진을 선명하게 하는 효과가 있습니다. -
포장지 및 포장박스
SS-SOL을 포함한 경우 내구성과 방습성을 향상시킵니다. -
셀로판 필름
SS-SOL과 글리세롤, Monostealate등의 왁스재료와 혼합한 수조에 필름을 담궈놓는 공정을 통하여 필름끼리 달라붙는 현상을 방지할 수 있습니다.
06 주조

-
정밀주조
SS-SOL은 세라믹 재료들과 강한 접착력을 가지고 있으므로 Ethyl silicate 대체품으로 사용하여 비용을 절감하고 작업환경의 개선효과 등이 가능합니다. 또한 소립자의 SS-SOL을 사용하는 경우 세라믹 재료의 강도를 개선할 수 있습니다. -
착색
휘발성 착색재료의 주형 강도증강을 위해 기존에 사용된 메탄올 대신 SS- SOL을 사용한 경우 전성이 향상되고 주형의 강도가 증대되며 주조의 표면을 매끄럽게 처리할 수 있습니다. -
CO2형 반응 촉진제
CO₂공정에서 규산소다와 탄산가스의 반응성이 낮으므로 그 공정에서 SS-SOL을 사용한 경우 반응속도가 올라가고, 작업성이 현저하게 증대됩니다.
07 세라믹 내화물

SS-SOL은 내열성 및 산에 견디는 성질과 응집력으로 인해 새로운 내화물질로 주목 받 고 있습니다. SS-SOL은 절연물질의 응집제로도 이용이 가능하며 탄소물에
이용될 때는 표면의 부식을 방지할 수 있습니다. 또한 철강제품 또는 내화물의 표면에 SS-SOL 단독 혹은 내화물질의 분말을 혼합하여 코팅을 하면 항열 항부식 효과를
가져옵니다.
이용될 때는 표면의 부식을 방지할 수 있습니다. 또한 철강제품 또는 내화물의 표면에 SS-SOL 단독 혹은 내화물질의 분말을 혼합하여 코팅을 하면 항열 항부식 효과를
가져옵니다.
08 무기섬유

-
유리섬유
SS-SOL을 유리섬유에 사용하는 경우, 섬유표면이 평평하고 미끄러워 생기는 섬유상호간의 들뜸현상을 방지할 수 있으며 섬유와 수지사이의 수분층 발생을 감소시킵니다. -
세라믹섬유
내열성, 내구성이 필요로 되는 세라믹 섬유의 생산 시 SS-SOL을 사용할 수 있습니다. 특히 세라믹 섬유를 이용한 WET FELT, SLEEVE, LADLE, 종이 그리고 TAPE의 재료 생산 시 SS-SOL을 접착제로 사용하면 내열성과 내구성이 높은 제품을 생산할 수 있습니다.
09 페인트, 접착제

피막제 및 접착제로써 비닐 아세테이트, 아크릴 에스테르, 합성고무등의 고분자와 SS- SOL을 사용함으로써 접착성, 견고성, 내마모성을 향상시킬 수 있습니다.
페인트의 기초 재료로 SS-SOL이 사용되면 콜로이달 실리카의 작용으로 필름에 발생하는 얼룩을 방지할 수 있습니다.
페인트의 기초 재료로 SS-SOL이 사용되면 콜로이달 실리카의 작용으로 필름에 발생하는 얼룩을 방지할 수 있습니다.
10 촉매

SS-SOL을 규산소다 대신 사용하게 되면 작업성이 현저하게 향상되며 제품의 표면적이 커지며, 촉매의 강도를 증가할 수 있습니다.
11 배터리

납축전지에서 콜로이달 실리카는 황산과 반응하여 겔화되고, 겔화된 전해액은 누수방지효과 및 배터리 성능향상을 나타낸다.
12 왁스

SS-SOL을 혼합한 왁스를 사용하면 광택효과를 상승시키고 미끄러짐을 방지할 수 있습니다.
13 Polishing

SS-SOL을 이용하여 화학적 연삭 작용 및 물리적 연마 작용을 원활히 작용할 수 있습니다. 실리콘 웨이퍼의 연삭 등 특수산업에서 사용됩니다.


