(주)에스켐텍
액상규산Liquid Sodium Silicate
일반적으로 "water glass"라 불린다.
분자식 : Na2O·nSiO2·xH2O
건식(간접법)과 습식방식(직접방법) 당사는 간접법으로 제조
용도 : 분자가루의 순수물질로서 Silica gel, 응고 Silica, 산업용 렌즈, 주조, 목제, 종이판사의 고착제, 세제의 보조물,
· 산업용 액화 Sodium Silicate는 무색, 약한 투명한 색상 또는 반투명 시럽같은 용액
· 산업용 고체 Sodium Silicate는 무색, 약한 투명한 색상, 반투명미립 또는 고체의 유리형태의 덩어리
분자식 : Na2O·nSiO2·xH2O
건식(간접법)과 습식방식(직접방법) 당사는 간접법으로 제조
용도 : 분자가루의 순수물질로서 Silica gel, 응고 Silica, 산업용 렌즈, 주조, 목제, 종이판사의 고착제, 세제의 보조물,
· 산업용 액화 Sodium Silicate는 무색, 약한 투명한 색상 또는 반투명 시럽같은 용액
· 산업용 고체 Sodium Silicate는 무색, 약한 투명한 색상, 반투명미립 또는 고체의 유리형태의 덩어리
01 일반적 특성
- 과잉 희석하면 H2O에 가깝다.
- 알칼리의 양이 감소하면 Silica Gel 또는 Silica Sol의 성질을 띤다.
- H2O의 양이 감소하면 無水 琉璃의 성질에 가깝다.
- 알칼리의 양이 증가하면 NaOH의 성질에 가깝다.
(단, 이경우 위의 4가지 특성중 다른 물질의 성질을 완전히 배제하는 일은 불가능하므로
알칼리성이 아닌 규산 나트륨 또는 규산이 없는 규산 나트륨은 전혀 생각할 수 없다.)
- 알칼리의 양이 감소하면 Silica Gel 또는 Silica Sol의 성질을 띤다.
- H2O의 양이 감소하면 無水 琉璃의 성질에 가깝다.
- 알칼리의 양이 증가하면 NaOH의 성질에 가깝다.
(단, 이경우 위의 4가지 특성중 다른 물질의 성질을 완전히 배제하는 일은 불가능하므로
알칼리성이 아닌 규산 나트륨 또는 규산이 없는 규산 나트륨은 전혀 생각할 수 없다.)
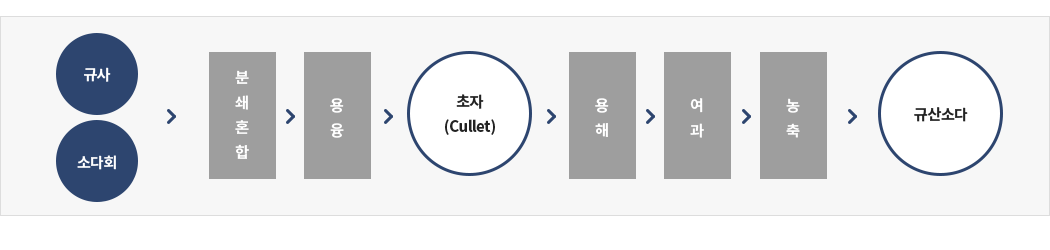
02 제조공정
-
간접법(건식법)
① 탄산 나트륨(소다회)과 규사를 배합비에 맞추어 혼합하여 용융로에 투입하여 용융
② 공기중에 냉각(cullet)
③ 용해관에 물과 함께 넣고 가온 가압시켜 용해
④ 여과기로 불순물을 제거
⑤ 정해진 농도에 따라 농축시켜 제품화. Na2CO3 + nSiO2 → Na2O·nSiO2 + CO2
-
직접법(습식법)
① NaOH와 규사분말을 조성비에 맞추어 직접 용해관에 투입
② 가압 증기와 반응시킨 후 여과 농축하여 제품화

03 물리화학적특성
-
1. 조성
일반적으로 규산 나트륨 용액은 Na2O·nSiO2·xH2O로 표시된다. 수화상태의 고분자 Silica 산은 OH 나 ONa와 결합한 -O-Si-O 결합을 형성하지만 결합의 완전한 수화현상 때문에 Na 와의 결합은 매우 약하다. n은 Mole비(M.R)를 나타낸다.
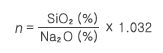
-
2. 비중
온도가 상승하면 비중은 하강한다. Baume도는 20℃에서 측정한 수치로 표시. Be(20℃) = [(20-t) × 0.04] 액상 규산 나트륨의 조성과 물리적 성질(20℃)Na2O SiO2(%) Mole Ratio Baume Viscosity(CP) 19.7 31.2 1.6 58.0 3,000 14.7 29.4 2.1 50.0 122 13.9 33.4 2.4 52.0 640 10.6 26.9 2.6 42.0 23 11.2 31.9 2.9 47.0 250 10.3 30.9 3.1 44.6 150 9.1 29.2 3.3 41.5 75 8.8 29.1 3.4 41.0 110 6.6 25.3 4.0 34.0 75 -
3. 점도(Viscosity)
동일한 M.R에서는 고형분이 증가할수록 점도가 높아진다. 고형분이 동일한 경우 M.R이 낮을수록 점도가 낮아진다. M.R이 낮더라도 비중이 증가하면 점도가 높아진다. 온도가 증가하면 점도가 낮아진다. 점도가 동일한 경우 M.R이 낮을수록 비중은 증가한다. -
4. pH
pH 11.5~13.0에서 완충현상 M.R이 낮을수록( 함량이 높을수록) pH는 높아진다. -
5. 빙결점
M.R 3.3, 고형분 함량 38%에서 액상 규산 나트륨(3호)의 빙결점은 -2.3℃ M.R 2.0, 고형분 함량 54%인 경우 빙결점은 -32℃ 빙결된 액상 규산 나트륨은 사방정계 결정을 형성 하지만 가열해 녹이면 다시 원상태의 물성 으로 환원한다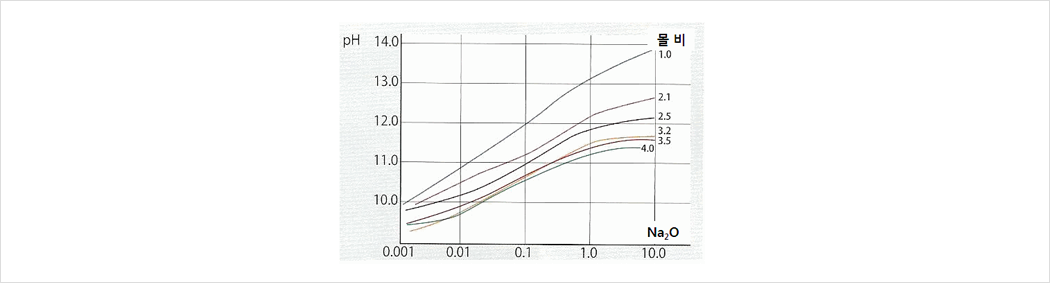
-
6. 액상 규산 나트륨을 산으로 중화하면 규산이 순간적으로 분리하여 Gel을 생성한다. 또한 물의 방출에 의해 수축이 시작되는 현상이 일어나 규산 나트륨의 성질을
잃어 버린다.
7. 액상 규산 나트륨의 100% 탈수는 1,000℃ 이상에서 가능하며 또한 탈수가 일어나더라도 규산 나트륨의 기본적 성질은 변하지 않는다.
8. Al, Ca, Ba 등의 금속은 규산 나트륨에 작용하여 규산 금속의 수산화물 및 물에 의해 여러 가지 색을 띠는 조성의 침전을 생성한다.
9. 액상 규산 나트륨에 알코올을 첨가하면 알코올의 탈수 작용에 의해 Gel을 생성한다.
04 취급상 주의사항
-
규산 나트륨은 독극물이 아닌 무해의 약품이다. 단, 식품첨가물이 아니므로 마시면 인체에 해롭다.
- 피부에 묻었을 때 물로 깨끗이 씻어야 한다.
- 눈에 들어갔을 경우 깨끗한 물로 세안하고 붕산수로 씻은 후 안약을 넣어야 한다.
- 상처 부위에 닿지 않도록 주의한다.
- 의복에 묻어 건조될 경우 잘 떨어지지 않고 의복이 탈색될 우려가 있으므로 건조되기 전에 가능한 빨리 미지근한 물로써 깨끗이 씻어야 한다.
05 액상 규산 나트륨의 용도
| 용도 | mole 비 | 주요특성 | |||||
|---|---|---|---|---|---|---|---|
| 접 착 | Silicic산의 발생원 | 화학 반응성 |
분산 유화성 |
중화제 비누화제 |
과산화물 안정제 |
||
| 종이상자 | 3.3 | ○ | |||||
| 용접봉 | 2.8~3.3 | ○ | |||||
| 페인트 | 3.3 | ○ | |||||
| 주물주강 | 2~2.7 | ○ | ○ | ||||
| 지질강화 | 3.3 | ○ | |||||
| 수처리 | 3.3 | ○ | |||||
| 무수 Silicic산 | 3.3 | ○ | ○ | ||||
| 내화물 | 3.3 | ○ | ○ | ||||
| 화학제품 | 2~3.3 | ○ | ○ | ||||
| 시 멘 트 | 2~3.3 | ○ | |||||
| 섬유공업 | 2~3.3 | ○ | ○ | ○ | |||
| 제지공업 | 2~3.3 | ○ | ○ | ○ | |||
| 세제비누 | 2~3.3 | ○ | ○ | ○ | |||

1. 주형 점결제
Gel을 형성하는 규산 나트륨의 특성을 이용한 것으로 M.R 2.1~3.2의 규산 나트륨 용액 사용.주물사에 규산 나트륨 용액을 첨가하고 탄산가스를 주입하여 Silica Gel을 형성하는 CO₂ 공정은 장치비와 인건비 절감,
적은 공간 차지, 악취가 발생하지 않아 널리 보급되고 있다.

2. 토양 강화제
토목,건축공사에 있어서 강력한 지수효과를 나타내는 방수제의 주원료로 사용.Grouting 분야에 있어서 약액 주입 공법의 발전으로 지반 구성이 복잡하고(점토사 또는 모래사로 되어있는 지반 등 견고하지 못한 지반)
지하수가 많이 포함된 지층에서의 지수처리, 지반개랑 등 액상 규산 나트륨을 주체로 한 약액 주입이 실시되고 있다.


3. 세제(비누, 합성세제)
세제의 助 Buidler로 작용하여 경수의 연화 작용을 한다. 액상 규산 나트륨 중의 알칼리가 직접 유리된 탄산과 화합하여 산성의 탄산염을분해하는 한편, 규석분이 점차 물중에 있는 칼슘이나 마그네슘염과 결합하여 매우 연한 Gel 상의 미세한 침전을 만들어 용액중에
부유함으로써 생긴다. 세제의 Buffer 작용 및 Suspension Emulsifying제로 이용되며, 세탁 助재질인 알루미늄, 아연 등의
부식방지 작용도 수행한다.

4. 무기 접착제
M.R 2.0~3.4의 액상 규산 나트륨은 쉽게 표면을 적시고 한계내의 침투를 일으키며 적당한 점도 등의 물성을 갖추고 있어 종이상자,석면제품 종이관 등의 접착에 이용된다. 또한 최근에는 내화 및 내열성 무기 도료, 건축용 무기도료, 환경 친화성 건축내장재의 접착제 등
다양한 용도에 널리 쓰이고 있다.

5. 제지, 펄프
액상 규산 나트륨은 종이의 脫墨製로서 또한 펄프 표백의 경우 과산화 수소의 안정제로 이용.
6. Cement
내산 내화를 요하는 시멘트 점결제로 이용되며 단열제 부착에도 이용된다.-
7. Silica Sol, Water Treatment
규산 나트륨을 중화하면 Silicic 산이 방출되어 고분자화하며 Silicic 산의 미립자를 형성하여 강한 음성을 갖는 Silica Sol이 형성된다.
이 Silica Sol은 원수와 폐수의 응집제로 이용된다. -
8. 화학 제품
규산나트륨을 산처리하여 촉매 흡착제로 널리 쓰이는 Silica Gel을 만들며, 역시 산처리하여 초기에 생기는 미립규산 분말은 화이트 카본이라 불리며
고무 충진제로 널리 이용된다. -
9. 그외 규산 금속 제조, 메타 규산 나트륨의 원료, 섬유, 요업등에 널리 이용되고 있다.
韓國工業規格 KSM 1415
항 목 1 종 2 종 3 종 4 종 비중 (20℃) 1.690 이상 1.590 이상 1.380 이상 1.260 이상 물불용분(%) 0.2 이하 0.2 이하 0.2 이하 0.2 이하 Na2O (%) 17 - 18 14 - 15 9 - 10 6 - 7 SiO2 (%) 36 - 38 34 - 36 28 - 30 23 - 25 Fe2O3 (%) 0.05 이하 0.05 이하 0.03 이하 0.03 이하


